|
This process reduces the atomic number by 1 and. > electron capture - Electron capture is a type of radioactive decay where the nucleus of an atom absorbs a K or L shell electron and converts a proton into a neutron. The electron converts a proton to a neutron in the nucleus. One example of electron capture involves beryllium-7. electron capture: capture of an electron by an unstable nucleus. 
Often the nucleus exists in an excited state as well and emits a gamma ray as it transitions to the ground state of the new nuclide. (We pronounce the name as "oh zhay", not "aw ger). Write equations for the following nuclear reactions. We call this ejected electron an Auger electron after one of its discoverers, Pierre Victor Auger. Science Chemistry Write equations for the following nuclear reactions. Iodine-131 is an example of a nuclide that undergoes decay: 53 131 I -1 0 e + 54 131 Xe or 53 131 I -1 0 + 54 131 Xe Beta decay, which can be thought of as the conversion of a neutron into a proton and a particle, is observed in nuclides with a large n:p ratio. All it does is carry away excess energy.Īnother electron may absorb excess energy and leave the atom. Beta () decay is the emission of an electron from a nucleus. The facts are that both EI and ECNI involve the gas-phase analyte and an electron however, the energy of the electron is very different in EI and ECNI. ECNI is a reaction occurring between an electron and a gas-phase analyte molecule. Many people omit the neutrino in the equation, because it has no mass or charge. Electrons are extremely light in weight so they do not contribute towards weight but they have a great role in chemical reactions. CI (regardless of the sign of the charge of the reagent ion or the product) is an ion/molecule reaction. The excess energy leaves as an X-ray photon and a neutrino. The inner shell is missing an electron.Īn outer shell electron then drops to a lower energy level to replace the missing electron. The atom stays neutral in charge, but it now exists in an excited state. The captured electron often comes from the K-shell because those electrons are closest to the nucleus. Since a proton becomes a neutron, the number of protons decreases by 1, but the atomic mass stays the same. The device was invented in 1957 by James Lovelock and is used in gas chromatography to detect trace amounts of chemical compounds in a sample. 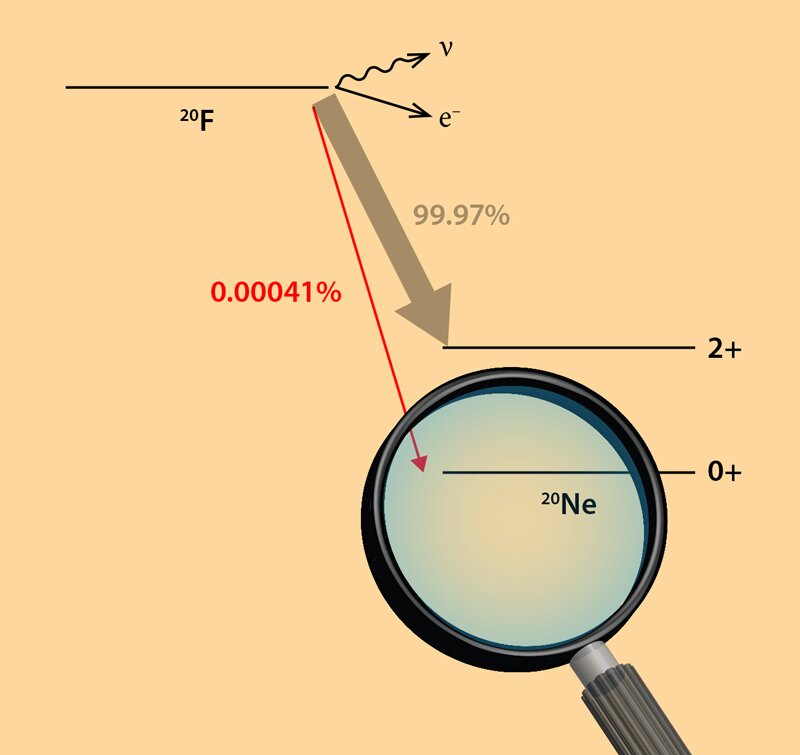
The electron combines with a proton to make a neutron and emits an electron neutrino.Įlectron capture is a major decay mode for isotopes with too many protons in the nucleus. An electron capture detector (ECD) is a device for detecting atoms and molecules in a gas through the attachment of electrons via electron capture ionization. An electron capture detector ( ECD) is a device for detecting atoms and molecules in a gas through the attachment of electrons via electron capture ionization.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
